“What is the difference between pap smears and HPV testing? This is a question that I frequently get asked as a specialist in Maternal Fetal Medicine.
In this article, I want to tell you everything you need to know when it comes to protecting yourself against cervical cancer, including the differences and similarities in pap smears and HPV testing.
Related article: The Difference Types of HPV: What You Should Know
What is the difference between pap smears and HPV testing?
PAP smears and HPV testing have the same purpose which is to screen for cervical cancer. Screening aims to detect precancerous changes, which, if not treated, may lead to cancer.
Pap smears and HPV screenings test for precancerous cells of the cervix. The cervix is the opening of the uterus (womb). It is part of a woman’s reproductive system.
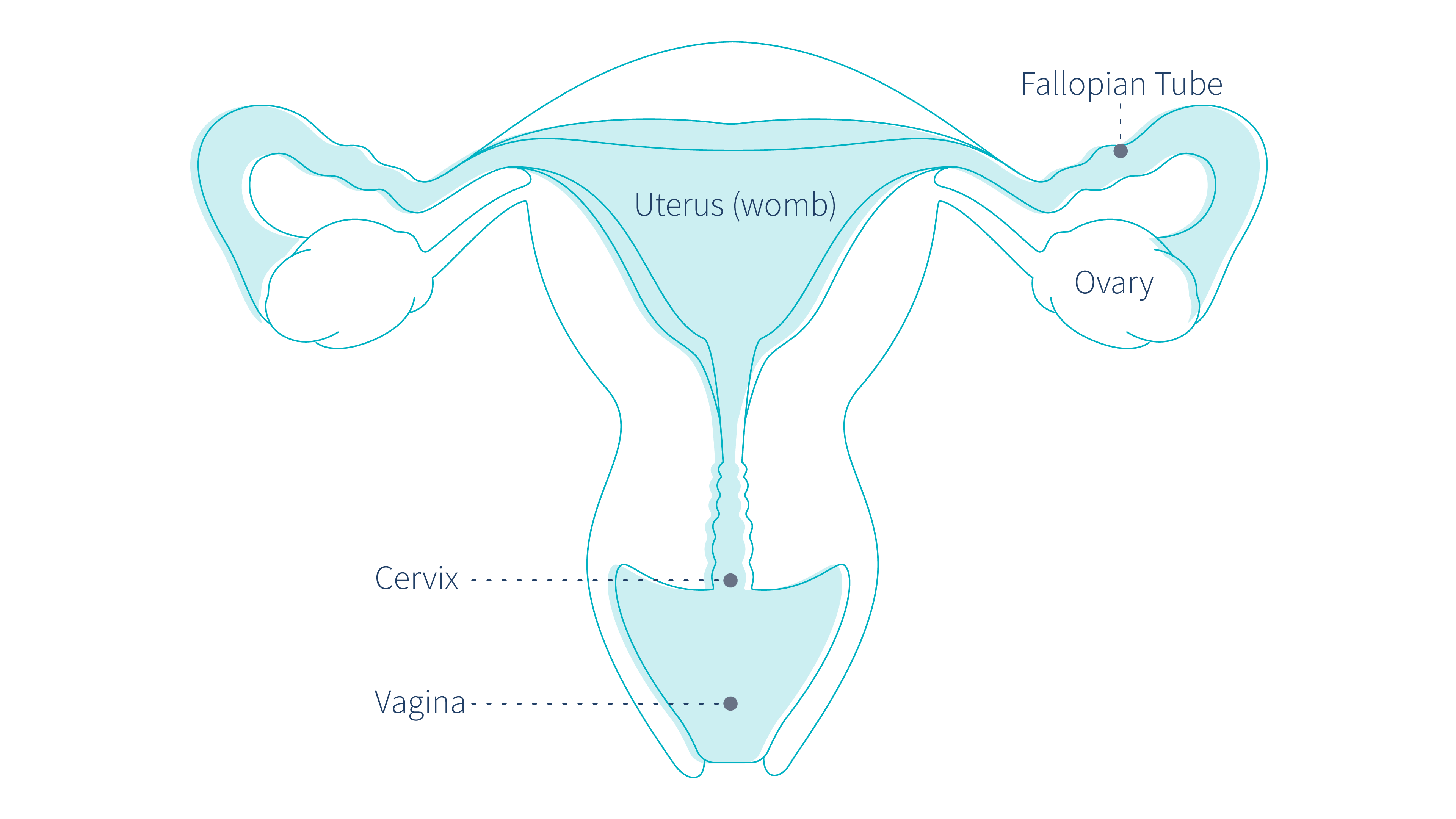
The difference in pap smears and HPV testing lies in what is being examined from the collection of cells.
Pap Smears
Pap smears looks for precancerous cell changes on the cervix that might become cervical cancer if not treated appropriately. The Pap test can also find cancer cells.
- A Pap smear result can be normal, abnormal or unclear.
- In a conventional Pap test, the cells are placed on a glass microscope slide and a fixative is added. In an automated liquid-based Pap cytology test, cervical cells collected with a brush or other instrument are placed in a vial of liquid preservative.
- The slide or vial is then sent to a laboratory for analysis.
HPV Testing
The HPV test detects DNA or RNA from certain high-risk types of HPV in samples of cells taken from the cervix. In other words, HPV testing looks for the presence of virus in the cervical cells.
- HPV tests can detect HPV infection often before cell abnormalities have occurred. An HPV test comes back as either positive or negative.
- A negative HPV test means you do not have a HPV type that is linked to cervical cancer.
- A positive HPV test means you do have an HPV type that may be linked to cervical cancer. This does not mean you have cervical cancer now.
Both PAP testing and HPV testing are performed by a healthcare professional. An instrument called a speculum is inserted into a woman's vagina to widen it so that the upper portion of the vagina and the cervix can be seen. A sample of cervical cells are taken using a wooden or plastic scraper and/or a cervical brush.
One advantage of liquid-based testing is that the same cell sample can also be tested for the presence of high-risk types of HPV, a process known as “Pap and HPV co-testing.”
In addition, liquid-based cytology appears to reduce the likelihood of an unsatisfactory specimen. An HPV test can also be performed separately from a PAP smear by obtaining a sending a sample of cells from the cervical canal to check for the presence of HPV virus.
Research on the differences in pap smears and HPV testing
There has been a large volume of research completed on the differences in pap smears and HPV testing. Cervical cancer screening tests are highly effective, but they are not completely accurate.
Sometimes a woman can be told that she has abnormal cells when the cells are actually normal (a false-positive result), or she can be told that her cells are normal when in fact there is an abnormality that was not detected (a false-negative result).
Both false negative and false positive test results are problematic. A false negative test result can cause a cancer to go undetected, whereas a false positive test result can result in unnecessary treatment which can have risks.
Cervical cancer screening has another limitation, caused by the nature of HPV infections. Because most HPV infections are transient and produce only temporary changes in cervical cells, overly frequent cervical screening could detect HPV infections or cervical cell changes that would never cause cancer.
Treating these abnormalities that would have gone away on their own can cause needless stress. In addition, follow-up tests and treatments can be uncomfortable, and some treatments that remove cervical tissue to treat abnormal cells, such as LEEP and conization, have the potential to weaken the cervix and may slightly increase the risk of premature delivery in pregnant women, depending on how much tissue is removed.
Furthermore, a small minority of cervical cancers are not caused by HPV, raising the possibility that HPV screening would miss women at risk for those rare cancers.
Women in their 20s are advised to get Pap smears, not HPV tests, because the virus is so common that most would test positive for infections that would most likely clear up on their own.
For women age 30 and older, Pap and HPV co-testing is less likely to miss an abnormality (i.e., has a lower false-negative rate) than Pap testing alone.
Therefore, a woman with a negative HPV test and normal Pap test has very little risk of a serious abnormality developing over the next several years.
In fact, researchers have found that, when Pap and HPV co-testing is used, lengthening the screening interval to 5 years still allows abnormalities to be detected in time to treat them, while also reducing the detection of HPV infections that would have gone away on their own.
Adding HPV testing to Pap testing may also improve the detection of glandular cell abnormalities, including adenocarcinoma of the cervix (cancer of the glandular cells of the cervix).
There is some evidence that Pap testing is not as good at detecting adenocarcinoma and glandular cell abnormalities as it is at detecting squamous cell abnormalities and cancers. One disadvantage of co-testing is that it results in higher costs.
In 2014 the FDA approved the first HPV test that can be used as a primary screening test for cervical cancer instead of a Pap smear for women age 25 and older. This test analyzes a sample of cells from the cervix for the presence of any of 14 HPV subtypes that increase the risk of cervical cancer.
An advantage of the HPV test is that the results are fully automated (that is, machine-read), whereas the Pap test relies in part on the expertise of medical personnel to detect cell abnormalities.
HPV testing identifies the presence of the HPV virus that is associated with cervical cancer.
The new approval was based on long-term findings from the ATHENA trial that included more than 47,000 women. The results showed that the HPV test used in the study performed better than the Pap test at identifying women at risk of developing severe cervical cell abnormalities.
A more recent study published in JAMA also demonstrated that among women undergoing cervical cancer screening, the use of primary HPV testing compared with Pap smear cytology testing, resulted in a significantly lower likelihood of high grade precancerous cervical changes at 48 months.
The FDA’s decision remains controversial. Some public-health and consumer groups oppose the HPV test as a stand-alone screening tool because they think its safety and effectiveness have not been sufficiently tested.
Among the concerns are that it could lead to over testing and overtreatment with more invasive procedures, especially in younger women, who are more likely to test positive for HPV, but who are at low risk for cervical cancer.
Primary HPV testing has not yet been incorporated into most current professional cervical cancer screening guidelines. All medical professional groups recommend that average-risk women get screened at regular intervals between the ages of 21-65. Women at higher risk for cervical cancer may require more frequent screening/monitoring. Most medical professional organizations recommend that:
-
Women Age 21 to 29: be screened every 3 years with Pap testing, regardless of age of onset of sexual activity or other risk factors.
-
Women Age 30 to 65: be screened every 5 years with Pap AND HPV testing (co-testing) OR every 3 years with PAP testing.
Related article: Can You Have Sex With HPV?
What is the future for cervical cancer screening?
Several new tests are currently in development that can improve the evaluation of HPV-positive women.
One test that is still being evaluated but is not yet available is based on the detection of two proteins – p16 and Ki-67- which indicate an active HPV infection.
Checking cervical cells for the presence of P16/Ki-67 proteins was found to be more accurate at predicting whether an HPV-positive women would go on to develop cervical precancer within 5 years compared to Pap testing.
Additionally, researchers at the NCI have developed an artificial intelligence approach called automated visual evaluation (AVE) that can analyze digital images of a cervix and accurately identify precancerous changes. The images can be taken with a cell phone or similar device with a camera.
Another improvement that may be of particular importance where healthcare resources are limited is the ability to do self-sampling.
A clinical study conducted among more than 180,000 women in the Netherlands found that HPV testing done on cervical samples collected by women themselves had similar accuracy to that of clinician-collected samples for the detection of moderately to severely abnormal cervical cells.
In August 2018, the U.S. Preventive Services Task Force (USPSTF) released updated guidelines about screening for cervical cancer. The recommendations, based on a review of the latest research, say that average-risk women ages 30 to 65 can be screened for cervical cancer via any of three options:
- A Pap test every three years
- An HPV test every five years
- Both tests (co-testing using cervical cells collected at the same time) every five years
The Task Force does not recommend one option over the other, instead stating that the goal should be that “women receive adequate screening, regardless of which strategy is used.”
Because recommendations continue to change as new studies become available, it’s important to talk to your health care provider about which screening option is best for you.
The bottom line is that, unlike most cancers, there are excellent screening tests for cervical cancer.
The most important thing is to get screened on a regular basis in knowing your risk of cervical cancer.









